Why Xocova beats Paxlovid
I recently wrote a viral thread on X about Xocova, which is sometimes called the “Japanese Paxlovid”. It’s been approved in Japan since 2022 for treating COVID-19, but it’s never been approved in the US due to the FDA having gripes with some aspects of the company’s clinical trials.1
A head-to-head trial in The Lancet shows Xocova is as effective as Paxlovid in terms of reducing symptoms and fever in patients with COVID-19. Now data published in The New England Journal of Medicine shows the drug is highly effective when used prophylactically to prevent infection.
After studying both drugs, I concluded there would be major benefits to having Xocova approved in the US. That’s why I started a petition on change.org urging the FDA to approve the drug.
Xocova for post-exposure prophylaxis (PEP)
In March this year the PMDA (Japan’s equivalent to the FDA) approved Xocova for post-exposure prophylaxis (PEP). The idea here is that if someone tests positive for COVID-19, all of their family members and close contacts can get a prescription for the drug.
Studies show that when a household contact gets COVID-19, about 20-40% of housemates become infected. Given the significant risks of COVID-19 infection, and given the fact it is hard to mask properly and consistently, there is need for antivirals that can be taken prophylactically.
Shionogi, the maker of Xocova, ran an N=2,041 Phase III trial where they gave Xocova to household contacts within 72 hours. After 10 days, Xocova cut COVID-19 risk by 67%:
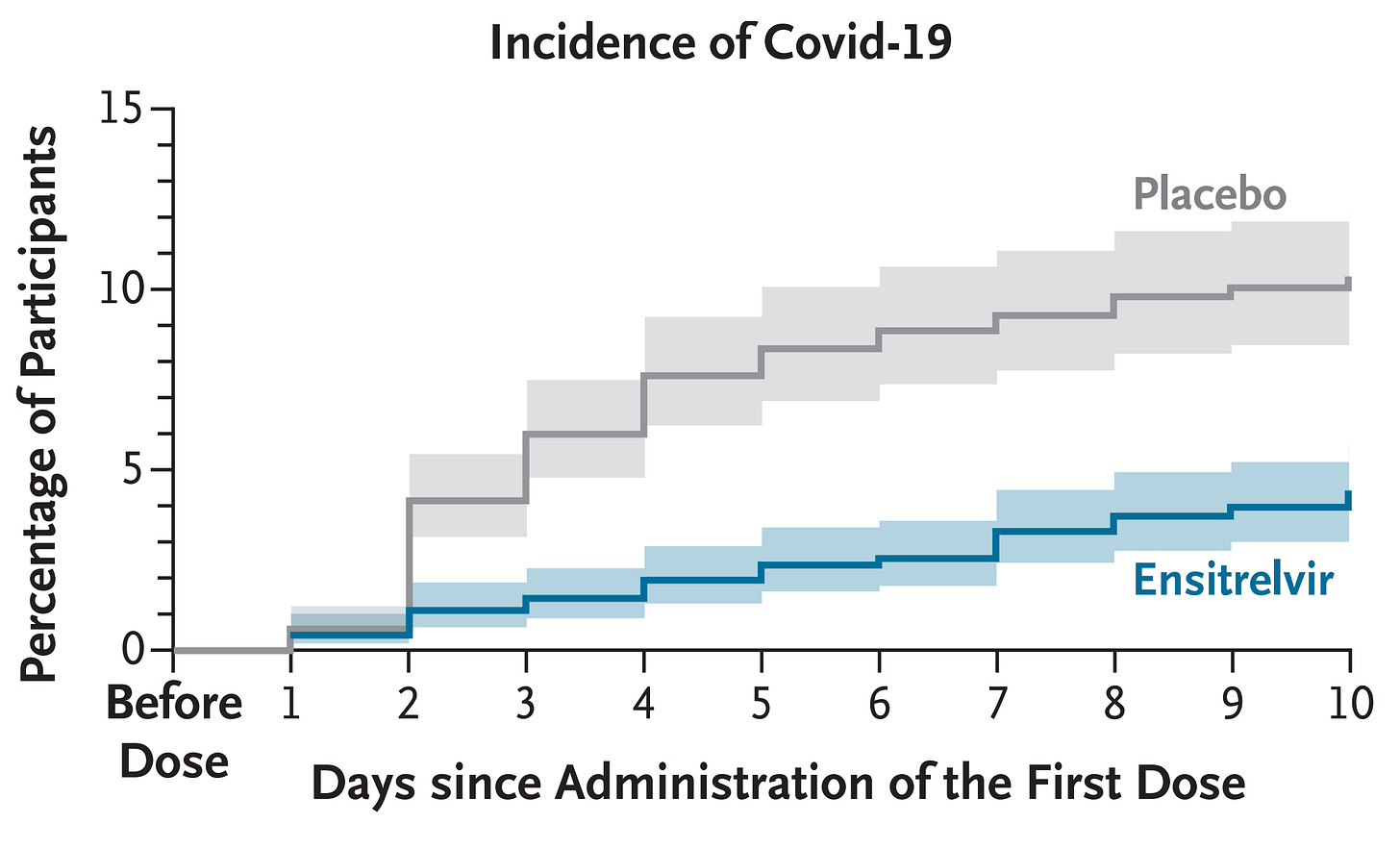
(Infection was determined by PCR testing, which is very sensitive, even asymptomatic cases would be counted.)
Xocova started a rolling submission to the FDA about a year ago to have Xocova authorized in the US for prophylactic use. From what I can tell, the application materials were completed about nine months ago. The FDA has set an “action date” of June 16th.
When Pfizer ran an N=2,736 trial on Paxlovid for post-exposure prophylaxis, they found a five day course cut risk by 32%, but that result was not statistically significant.
Why is Xocova better than Paxlovid for PEP?
A wealth of other data shows the two drugs are about equally effective at treating COVID-19 — so why did the Xocova PEP trial succeed while the Paxlovid PEP trial failed?
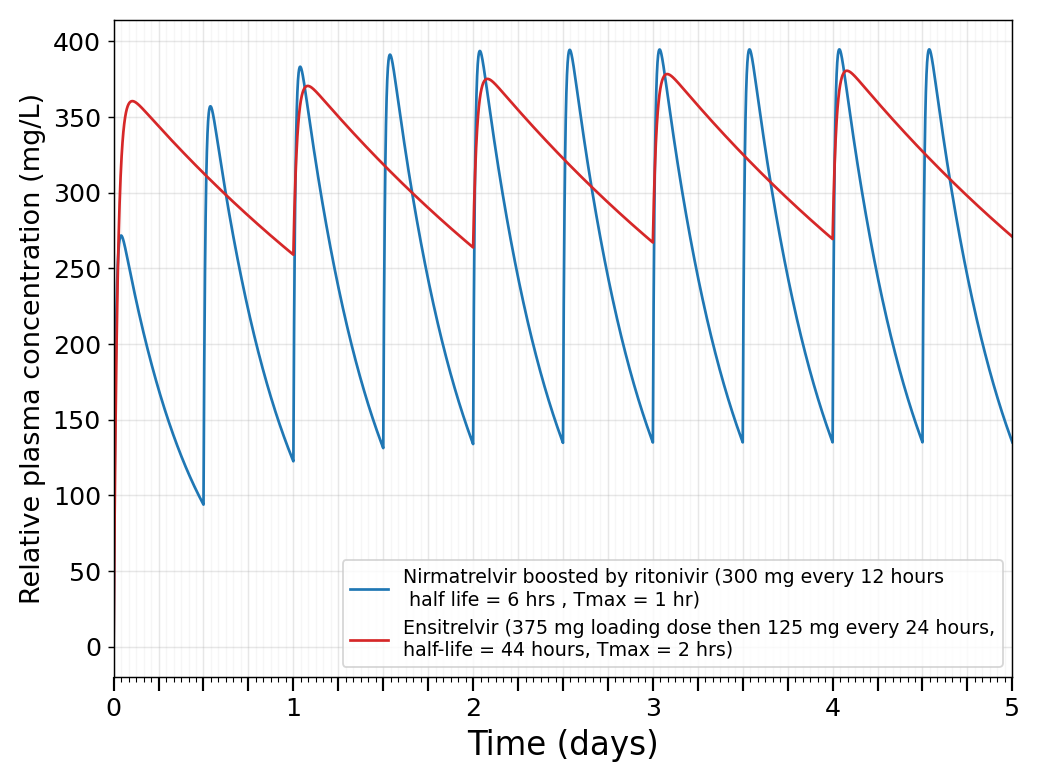
I think it has to do with the pharmacokinetics of the two drugs. The active ingredient in Paxlovid is Nirmatrelvir. The other ingredient is Ritonavir, which boosts Nirmatrelvir’s half-life. However, it only boosts it up to ~6-8 hours, so Paxlovid is dosed twice daily.
Xocova, on the other hand, has a long half life of 44 hours. Additionally, the company prescribes an initial loading dose of 375 mg, followed by 125 mg maintenance doses afterwards once a day for four days.
As a result, the concentration of Xocova (red) shoots up very quickly and remains high afterwards:
By contrast, the concentration of Paxlovid (blue) takes 24 hours to reach a steady state profile (follow the peaks on the blue lines - they take 24 hours to max out).
For anyone wondering, the binding affinity of the two drugs is almost identical in vitro (IC50=49 nM for Ensitrelvir vs IC50=44 nM for Nirmatrelvir in a head-to-head assay). So, for each mg of drug delivered into cells we can expect them to do about the same amount of work.
Because of Paxlovid’s short half-life, it dips to dangerously low levels every 12 hours. If you are exposed to COVID-19 during one of those troughs, the virus can start replicating and establish a foothold.
Taking these drugs as early as possible is really important
There’s another difference between the two trials that might be important. In the Xocova PEP trial, the drug was given within within 72 hours, while in the Paxlovid PEP trial, it was within 96 hours. Timing is really important here, and I think that one extra day could make a huge difference. In fact, I think it’s not implausible to think that if Xocova is taken within 24 hours of finding out a household contact has COVID-19, it lowers risk by 80% or more. Having the drug on hand in advance so it can be taken immediately is the best approach to using Xocova or any other prophylactic antiviral.
Why “Xocova rebound” isn’t a thing
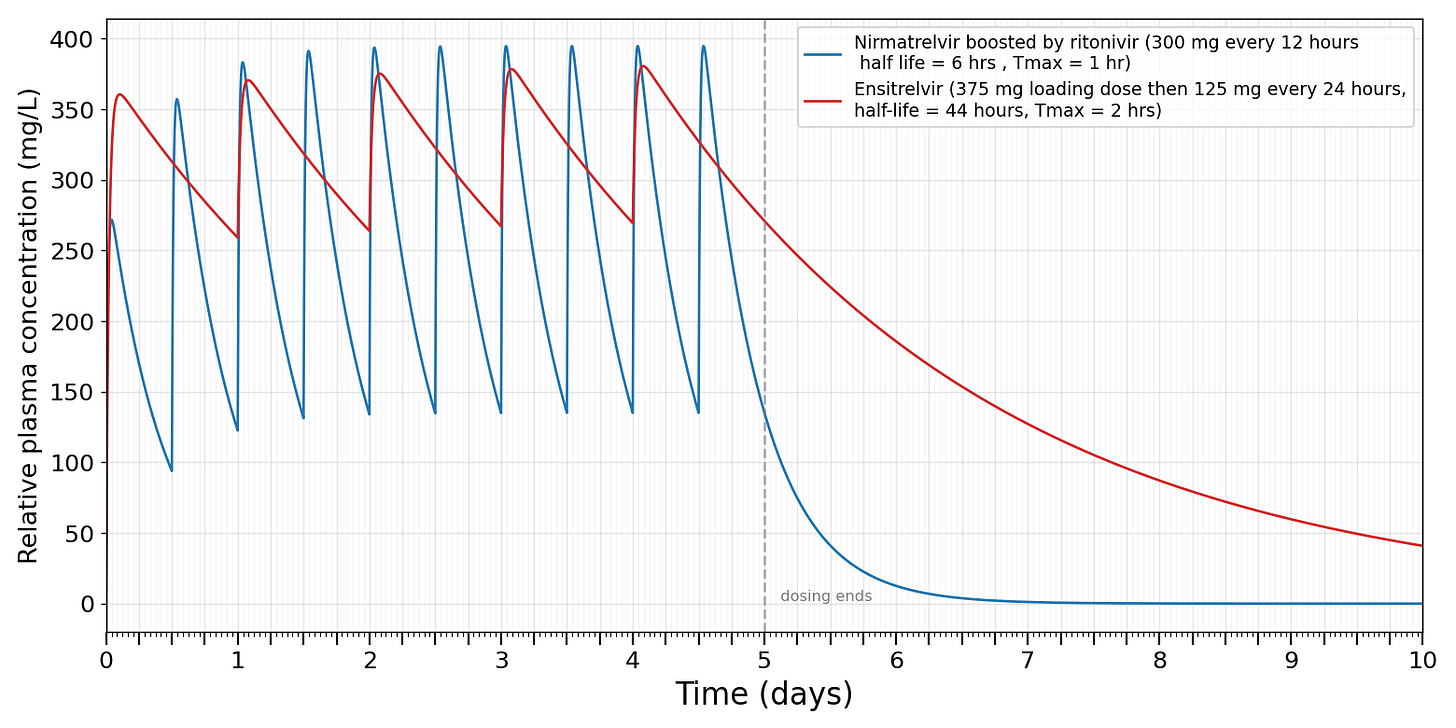
Xocova and Paxlovid are both prescribed in five day courses. Unfortunately, about 15% of people have a resurgence of their illness after they complete a five day course of Paxlovid, a phenomena called “Paxlovid rebound”. (Personally, I experienced minor Paxlovid rebound during my first time getting COVID-19, so when I got it a second and third time I made a point to take a 10 day course.)
I think this is pretty clearly related to Xocova’s long half life of 44 hours — the drug sticks around longer after the five day course ends:
Other benefits of Xocova
In addition to what we just discussed, there are other reasons Xocova is a better drug:
Paxlovid contains ritonavir, a liver enzyme inhibitor that inhibits CYP3A4. Unfortunately, ritonavir also boosts concentration of many common drugs (namely anti-psychotics, statins, antiplatelet drugs). These drug interactions can be hard to manage. Existing drugs have to be stopped, or the patient risks unwanted side effects. Important note (added 5/19): after publication I found out that Ensitrelvir is also a CYP3A4 inhibitor, to a significant degree, although it seems it isn’t as strong as ritonavir.
In trials run by Pfizer, about 6% of people who take Paxlovid report persistent unpleasant taste in their mouth, sometimes described as metallic or “soapy”. (The actual rate might be substantially higher - a trial where people used Paxlovid for 15 days found 60% reporting.) This generally doesn’t stop people from taking the drug, but it’s an unpleasant side effect you can avoid with Xocova. To be fair, Xocova does have its own side effects, the most notable being a temporary increase in blood triglycerides.
People with an allergy to polyethylene glycol (PEG) can’t take Paxlovid, since PEG is used in the tablet’s film, but they can take Xocova. This is only a tiny fraction of the population (<1%), but it’s just one more reason Xocova would be valuable as an option in the US.
Viral resistance to Xocova vs Paxlovid
Another interesting thing to consider is how different strains of SARS-CoV-2 are developing resistance to these two drugs. The two drugs both target the same enzyme (SARS-CoV-2 main protease), so one might worry that the virus could evolve resistance to both.
Data from 2022-2023 shows several strains that have resistance to one drug or the other, but only one strain that has exhibits some resistance to both. (This data is from Rueben Harris’s lab and the Midwest Antiviral Drug Discovery Center, whose entire funding was cut last year 😔.) The molecular structure of the two drugs is quite different, so that may be why it’s hard to evolve resistance to both.
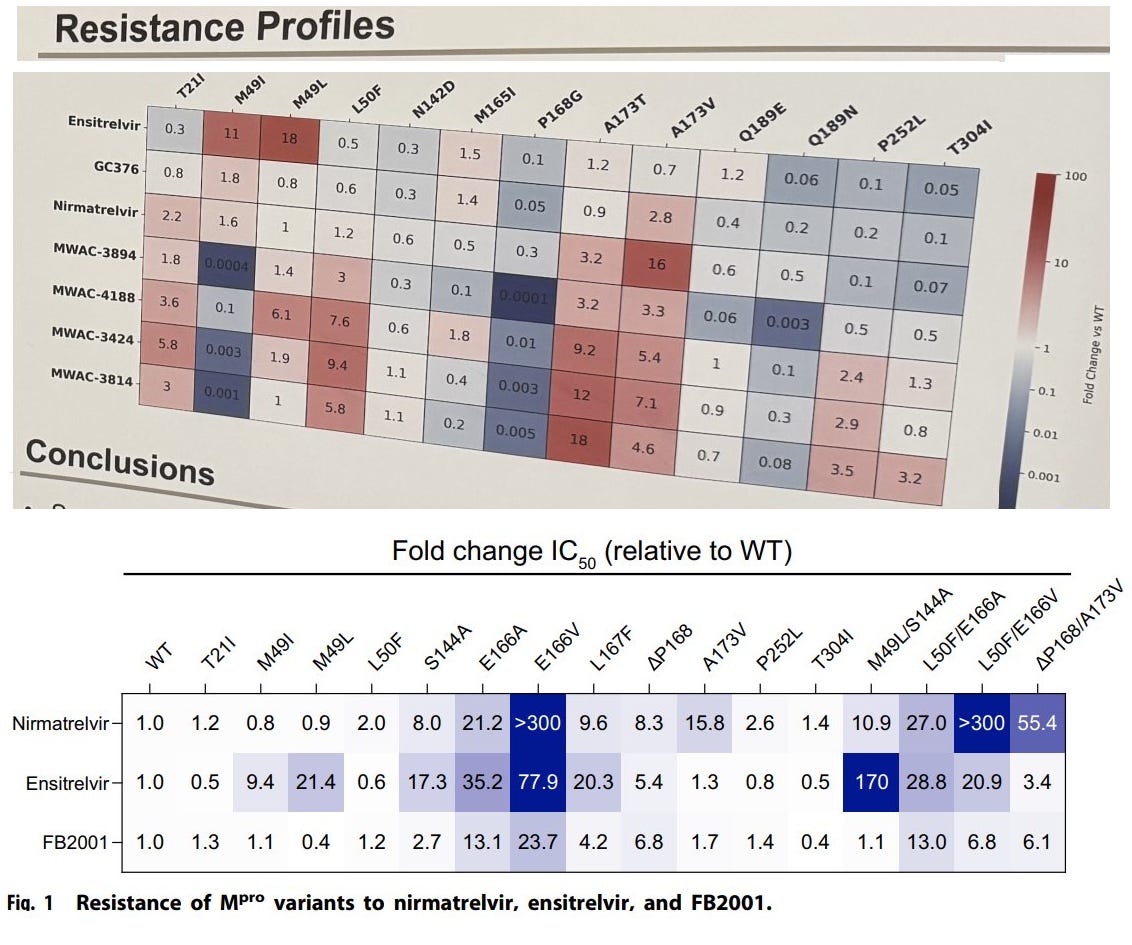
Interestingly, in 2025 researchers discovered a particular mutation which confers resistance to Xocova but at the same time increases the virus’s susceptibility to Paxlovid. The authors suggest “alternating or sequential antiviral regimens could effectively mitigate resistant variant emergence and spread in clinical settings.”
It may be that the optimal approach is to alternate the two drugs - Xocova one day, then Paxlovid the next (always starting with Xocova on the first day). That’s just another reason for the FDA to approve Xocova! Sign the petition!
Viral resistance to Xocova vs Paxlovid
The brief story is that the company has done three major trials: SCORPIO-SR, SCORPIO-HR, and SCORPIO-PEP. It was approved in Japan after SCORPIO-SR in 2022, but the FDA had two issues with that trial - first, the population was not diverse (it was only Asian people) and more importantly, the FDA thought that since the trial happened during the Omicron wave, it was not representative of the currently circulating viral strains. In response, the company ran a second trial, called SCORPIO-HR. The trial showed large and statistically significant drops in viral load. The trial’s primary endpoint, however, was symptom reduction. It showed symptom reduction, but failed to achieve statistical significance, leading the FDA to essentially reject their application. (It’s important to note that a 2025 meta-analysis of six trials and a head-to-head trial with Paxlovid all confirm the efficacy of the drug.) In 2025 the company pivoted to applying for an approval for post exposure prophylaxis on the basis of the successful SCORPIO-PEP trial.


Nice!
When I was evaluating antivirals for a long covid startup a couple years back, I used the ratio of [unbound Cmin: IC50], which takes into account pharmacokinetics including plasma protein binding. I already liked XOCOva back then because of it!
Xocova: 12.8 (low) - 26.7 (high)
Paxlovid: 4.3 (low) - 30.0 (high)
Higher is better. And these used substantially worse IC50 values than yours, 300-500nMfor xocova vs. 16-74nM for nirmatrelvir.
Personally in a pandemic I think this ratio would be a very acceptable surrogate endpoint, and we would just need to prove safety.
I can share the spreadsheet I made back then if you like
It would be nice to feel better faster, but my #1 concern with SARS-CoV-2 is minimizing the risk of long covid. I'm especially interested in keeping all my IQ points. For my purposes, the headline finding is that Xocova definitely reduces the risk of long covid (including neurological symptoms) and Paxlovid definitely doesn't:
https://www.eatg.org/hiv-news/shionogi-presents-pivotal-ensitrelvir-fumaric-acid-phase-3-data-and-exploratory-long-covid-data
https://www.cidrap.umn.edu/covid-19/studies-show-mostly-poor-long-covid-protection-paxlovid
The FDA is causing vast amounts of human misery with this ridiculous foot-dragging on the so-called "Fast Track." What we need is new law forcing the FDA to recognize convincing trials conducted in other countries - and to stop them from citing arbitrary "diversity" variables or other irrelevant trial minutiae when they choose to unilaterally withhold effective medicines from the American people. It's like we learned nothing from the Tuskegee Study:
https://christharrington.substack.com/p/the-lost-lesson-of-tuskegee